Methylammonium lead halide
From Wikipedia, the free encyclopedia
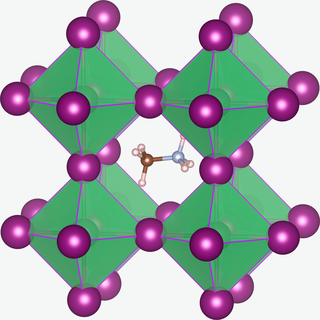
Methylammonium lead halides (MALHs) are solid compounds with perovskite structure and a chemical formula of [CH3NH3]+Pb2+(X−)3, where X = Cl, Br or I. They have potential applications in solar cells,[2] lasers, light-emitting diodes, photodetectors, radiation detectors,[3][4] scintillator,[5] magneto-optical data storage[6] and hydrogen production.[7]

Properties and synthesis
Summarize
Perspective
The first MALHs to be synthesized were the methylammonium derivatives [CH3NH3]SnX3 and [CH3NH3]PbX3. Their potential in the area of energy conversion wasn't realized until decades later.[8] In the [CH3NH3]PbX3 cubic crystal structure the methylammonium cation ([CH3NH3]+) is surrounded by PbX6 octahedra. The X ions are not fixed and can migrate through the crystal with an activation energy of 0.6 eV; the migration is vacancy assisted.[1] The methylammonium cations can rotate within their cages. At room temperature the ions have the CN axis aligned towards the face directions of the unit cells and the molecules randomly change to another of the six face directions on a 3 ps time scale.[9]
The solubility of MALHs strongly decreases with increased temperature: from 0.8 g/mL at 20 °C to 0.3 g/mL at 80 °C for [CH3NH3]PbBr3 in dimethylformamide. This property is used in the growth of MALH single crystals and films from solution, using a mixture of [CH3NH3]X and PbX2 powders as the precursor. The growth rates are 3–20 mm3/hour for [CH3NH3]PbI3 and reach 38 mm3/hour for [CH3NH3]PbBr3 crystals.[7]
The resulting crystals are metastable and dissolve in the growth solution when cooled to room temperature. They have bandgaps of 2.18 eV for [CH3NH3]PbBr3 and 1.51 eV for [CH3NH3]PbI3, while their respective carrier mobilities are 24 and 67 cm2/(V·s).[7] Their thermal conductivity is exceptionally low, ~0.5 W/(K·m) at room temperature for [CH3NH3]PbI3.[10]
Thermal decomposition of [CH3NH3]PbI3 gives methyl iodide (CH3I) and ammonia (NH3).[11]
[12]
- [CH3NH3]PbI3 → PbI2 + CH3I + NH3
Applications
MALHs have potential applications in solar cells, lasers,[13] light-emitting diodes, photodetectors, radiation detectors,[4] scintillator[5] and hydrogen production.[7] The power conversion efficiency of MALH solar cells exceeds 19%.[14][15]
Historic references
- Weber, Dieter (1978). "CH3NH3SnBrxI3-x (x = 0-3), ein Sn(II)-System mit kubischer Perowskitstruktur / CH3NH3SnBrxI3-x (x = 0-3), a Sn(II)-System with Cubic Perovskite Structure". Zeitschrift für Naturforschung B. 33 (8): 862–865. doi:10.1515/znb-1978-0809. ISSN 1865-7117.
- Weber, Dieter (1978-12-01). "CH3NH3PbX3, ein Pb(II)-System mit kubischer Perowskitstruktur / CH3NH3PbX3 , a Pb(II)-System with Cubic Perovskite Structure". Zeitschrift für Naturforschung B. 33 (12): 1443–1445. doi:10.1515/znb-1978-1214. ISSN 1865-7117. S2CID 93597007.*Grätzel, Michael (2014). "The light and shade of perovskite solar cells". Nature Materials. 13 (9): 838–842. Bibcode:2014NatMa..13..838G. doi:10.1038/nmat4065. ISSN 1476-1122. PMID 25141800.
See also
References
Wikiwand - on
Seamless Wikipedia browsing. On steroids.