Loading AI tools
Chemical compound From Wikipedia, the free encyclopedia
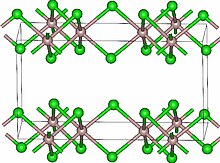
Lutetium(III) chloride or lutetium trichloride is the chemical compound composed of lutetium and chlorine with the formula LuCl3. It forms hygroscopic white monoclinic crystals[1] and also a hygroscopic hexahydrate LuCl3·6H2O.[6] Anhydrous lutetium(III) chloride has the YCl3 (AlCl3) layer structure with octahedral lutetium ions.[7]
 | |
| Names | |
|---|---|
| IUPAC name
Lutetium(III) chloride | |
| Other names
Lutetium chloride, lutetium trichloride | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.030.205 |
| EC Number |
|
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| LuCl3 | |
| Molar mass | 281.325 g/mol |
| Appearance | colorless or white monoclinic crystals |
| Density | 3.98 g/cm3 |
| Melting point | 925 °C (1,697 °F; 1,198 K)[1] |
| Boiling point | sublimes above 750°C[2] |
| soluble[3] | |
| Structure | |
| Monoclinic, mS16 | |
| C2/m, No. 12 | |
| Pharmacology | |
| License data | |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Irritant |
| GHS labelling:[4][5] | |
 | |
| Warning | |
| H315, H319, H335 | |
| P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Related compounds | |
Other anions |
Lutetium(III) oxide |
Other cations |
Ytterbium(III) chloride Scandium(III) chloride Yttrium(III) chloride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Lutetium-177, a radioisotope that can be derived from lutetium(III) chloride, is used in targeted cancer therapies.[8] When lutetium-177 is attached to molecules that specifically target cancer cells, it can deliver localized radiation to destroy those cells while sparing surrounding healthy tissue.[9] This makes lutetium-177-based treatments especially valuable for cancers that are difficult to treat with traditional methods, such as neuroendocrine tumors and prostate cancer.[10] Additionally, lutetium(III) chloride is used in scintillators, materials that emit light when exposed to radiation.[11] These scintillators are crucial in detectors for gamma rays and other high-energy particles, used in both medical diagnostics and in scientific research.[12]
Pure lutetium metal can be produced from lutetium(III) chloride by heating it together with elemental calcium:[13]
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.