
Lithium orotate
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Lithium orotate?
Summarize this article for a 10 year old
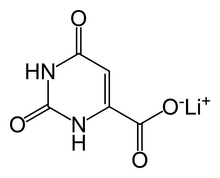
Lithium orotate (C5H3LiN2O4) is a salt of orotic acid and lithium. It is available as the monohydrate, LiC5H3N2O4·H2O.[1] In this compound, lithium is non-covalently bound to an orotate ion, rather than to a carbonate or other ion, and like other salts, dissociates in solution to produce free lithium ions. It is marketed as a dietary supplement, though it has been researched minimally between 1973–1986 to treat certain medical conditions, such as alcoholism[2] and Alzheimer's disease.
 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Monograph |
| Routes of administration | Oral |
| Legal status | |
| Legal status |
|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider |
|
| UNII | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.023.711 |
| Chemical and physical data | |
| Formula | C5H3LiN2O4 |
| Molar mass | 162.03 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
While lithium orotate is capable of providing lithium to the body,[3] like lithium carbonate and other lithium salts, there are no systematic reviews supporting the efficacy of lithium orotate and it is not approved by the U.S. Food and Drug Administration (FDA) for the treatment of any medical condition.