
Lithium diisopropylamide
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Lithium diisopropylamide?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
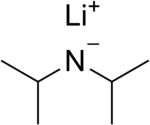
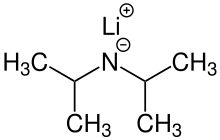
Lithium diisopropylamide (commonly abbreviated LDA) is a chemical compound with the molecular formula LiN(CH(CH3)2)2. It is used as a strong base and has been widely utilized due to its good solubility in non-polar organic solvents and non-nucleophilic nature. It is a colorless solid, but is usually generated and observed only in solution. It was first prepared by Hamell and Levine in 1950 along with several other hindered lithium diorganylamides to effect the deprotonation of esters at the α position without attack of the carbonyl group.[2]
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Lithium N-(propan-2-yl)propan-2-aminide | |
| Other names
LDA | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.021.721 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| LiN(CH(CH3)2)2 | |
| Molar mass | 107.1233 g/mol |
| Appearance | colourless solid |
| Density | 0.79 g/cm3 |
| Reacts with water | |
| Acidity (pKa) | 36 (THF)[1] |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
corrosive |
| Related compounds | |
Related compounds |
Superbases |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close