
Lead(II) chloride
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Lead(II) chloride?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Lead(II) chloride (PbCl2) is an inorganic compound which is a white solid under ambient conditions. It is poorly soluble in water. Lead(II) chloride is one of the most important lead-based reagents. It also occurs naturally in the form of the mineral cotunnite.
Quick Facts Names, Identifiers ...
 | |
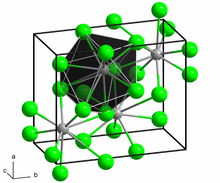 The crystal structure of PbCl2, in the unconventional crystallographic setting Pnam. This corresponds to the standard Pnma setting by switching the labels on the b and c axes. | |
| Names | |
|---|---|
| IUPAC names
Lead(II) chloride Lead dichloride | |
| Other names | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.028.950 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| PbCl2 | |
| Molar mass | 278.10 g/mol |
| Appearance | white odorless solid |
| Density | 5.85 g/cm3 |
| Melting point | 501 °C (934 °F; 774 K) |
| Boiling point | 950 °C (1,740 °F; 1,220 K) |
| 0.99 g/100 mL (20 °C)[1] | |
Solubility product (Ksp) |
1.7×10−5 (20 °C) |
| Solubility | slightly soluble in dilute HCl, ammonia; insoluble in alcohol Soluble in hot water as well as in presence of alkali hydroxide Soluble in concentrated HCl (>6M) |
| −73.8·10−6 cm3/mol | |
Refractive index (nD) |
2.199[2] |
| Structure[3] | |
| Orthorhombic, oP12 | |
| Pnma (No. 62) | |
a = 762.040 pm, b = 453.420 pm, c = 904.520 pm | |
Formula units (Z) |
4 |
| Thermochemistry | |
Std molar entropy (S⦵298) |
135.98 J K−1 mol−1 |
Std enthalpy of formation (ΔfH⦵298) |
-359.41 kJ/mol |
| Hazards[4] | |
| GHS labelling: | |
   | |
| Danger | |
| H302, H332, H351, H360, H372, H410 | |
| P201, P261, P273, P304+P340, P308+P313, P312, P391 | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LDLo (lowest published) |
140 mg/kg (guinea pig, oral)[5] |
| Related compounds | |
Other anions |
Lead(II) fluoride Lead(II) bromide Lead(II) iodide |
Other cations |
Lead(IV) chloride Tin(II) chloride Germanium(II) chloride |
Related compounds |
Thallium(I) chloride Bismuth chloride |
| Supplementary data page | |
| Lead(II) chloride (data page) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
