
Lanthanum hexaboride
Chemical compound / From Wikipedia, the free encyclopedia
Lanthanum hexaboride (LaB6, also called lanthanum boride and LaB) is an inorganic chemical, a boride of lanthanum. It is a refractory ceramic material that has a melting point of 2210 °C, and is insoluble in water and hydrochloric acid. It is extremely hard, with a Mohs hardness of 9.5.[1] It has a low work function and one of the highest electron emissivities known, and is stable in vacuum. Stoichiometric samples are colored intense purple-violet, while boron-rich ones (above LaB6.07) are blue. Ion bombardment changes its color from purple to emerald green.[2] LaB6 is a superconductor with a relatively low transition temperature of 0.45 K.[3]
Quick Facts Names, Identifiers ...
 | |
| Names | |
|---|---|
| Other names
Lanthanum boride | |
| Identifiers | |
| ChemSpider |
|
| ECHA InfoCard | 100.031.379 |
| EC Number |
|
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| Properties | |
| LaB6 | |
| Molar mass | 203.78 g/mol |
| Appearance | intense purple violet |
| Density | 4.72 g/cm3 |
| Melting point | 2,210 °C (4,010 °F; 2,480 K) |
| insoluble | |
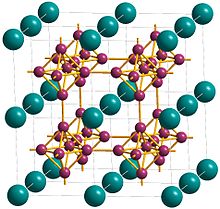
| Structure | |
| Cubic | |
| Pm3m ; Oh | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close


