
Ketoconazole
Antifungal chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Ketoconazole?
Summarize this article for a 10 year old
Ketoconazole, sold under the brand name Nizoral among others, is an antiandrogen, antifungal, and antiglucocorticoid medication used to treat a number of fungal infections.[11] Applied to the skin it is used for fungal skin infections such as tinea, cutaneous candidiasis, pityriasis versicolor, dandruff, and seborrheic dermatitis.[12] Taken by mouth it is a less preferred option and only recommended for severe infections when other agents cannot be used.[11] Other uses include treatment of excessive male-patterned hair growth in women and Cushing's syndrome.[11]
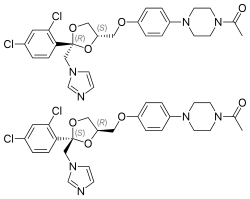 (2R,4S)-(+)-ketoconazole (top) (2S,4R)-(−)-ketoconazole (bottom) | |
 Ball-and-stick model of (2R,4S)-(+)-ketoconazole | |
| Clinical data | |
|---|---|
| Pronunciation | /ˌkiːtoʊˈkoʊnəˌzoʊl, -zɒl/[1][2] |
| Trade names | Nizoral, others |
| Other names | R-41400; KW-1414 |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682816 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | By mouth (tablets), topical (cream, shampoo, solution) |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | By mouth: 37–97%[8] |
| Protein binding | 84 to 99% |
| Metabolism | Extensive liver (predominantly oxidation, O-dealkylation) |
| Metabolites | N-deacetyl ketoconazole |
| Elimination half-life | Biphasic |
| Excretion | Bile duct (major) and kidney[9] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider | |
| UNII | |
| KEGG |
|
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.059.680 |
| Chemical and physical data | |
| Formula | C26H28Cl2N4O4 |
| Molar mass | 531.43 g·mol−1 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture[9][10] |
| |
| |
| | |

Common side effects when applied to the skin include redness.[12] Common side effects when taken by mouth include nausea, headache, and liver problems.[11] Liver problems may result in death or the need for a liver transplantation.[11][13] Other severe side effects when taken by mouth include QT prolongation, adrenocortical insufficiency, and anaphylaxis.[11][13] It is an imidazole and works by hindering the production of ergosterol required for the fungal cell membrane, thereby slowing growth.[11]
Ketoconazole was patented in 1977 by Belgian pharmaceutical company Janssen, and came into medical use in 1981.[14] It is available as a generic medication and formulations that are applied to the skin are over the counter in the United Kingdom.[12] In 2021, it was the 161st most commonly prescribed medication in the United States, with more than 3 million prescriptions.[15][16] The formulation that is taken by mouth was withdrawn in the European Union and in Australia in 2013,[17][18] and in China in 2015.[19] In addition, its use was restricted in the United States and Canada in 2013.[18]