
Iron(II) carbonate
Chemical, compound of iron carbon and oxygen / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Iron(II) carbonate?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
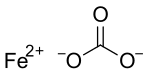
Iron(II) carbonate, or ferrous carbonate, is a chemical compound with formula FeCO
3, that occurs naturally as the mineral siderite. At ordinary ambient temperatures, it is a green-brown ionic solid consisting of iron(II) cations Fe2+
and carbonate anions CO2−
3.[5]
Quick Facts Names, Identifiers ...
 | |
| Names | |
|---|---|
| Other names
ferrous carbonate | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.008.418 |
| E number | E505 (acidity regulators, ...) |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| FeCO3 | |
| Molar mass | 115.854 g/mol |
| Appearance | white powder or crystals |
| Density | 3.9 g/cm3[1] |
| Melting point | decomposes |
| 0.0067 g/L;[2] Ksp = 1.28 × 10−11 [3] | |
Solubility product (Ksp) |
3.13×10−11[4] |
| +11,300·10−6 cm3/mol | |
| Structure | |
| Hexagonal scalenohedral / Trigonal (32/m) Space group: R 3c, a = 4.6916 Å, c = 15.3796 Å | |
| 6 | |
| Related compounds | |
Other anions |
iron(II) sulfate |
Other cations |
copper(II) carbonate, zinc carbonate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close