
Hypochlorite
An anion / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Hypochlorite?
Summarize this article for a 10 year old
In chemistry, hypochlorite, or chloroxide is an anion with the chemical formula ClO−. It combines with a number of cations to form hypochlorite salts. Common examples include sodium hypochlorite (household bleach) and calcium hypochlorite (a component of bleaching powder, swimming pool "chlorine").[1] The Cl-O distance in ClO− is 1.69 Å.[2]
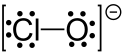 | |
 | |
| Names | |
|---|---|
| IUPAC name
Hypochlorite | |
| Systematic IUPAC name
Chlorate(I) | |
| Other names
Chloroxide | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.235.795 |
| 682 | |
PubChem CID |
|
| UNII | |
| UN number | 3212 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| Conjugate acid | Hypochlorous acid |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
The name can also refer to esters of hypochlorous acid, namely organic compounds with a ClO– group covalently bound to the rest of the molecule. The principal example is tert-butyl hypochlorite, which is a useful chlorinating agent.[3]
Most hypochlorite salts are handled as aqueous solutions. Their primary applications are as bleaching, disinfection, and water treatment agents. They are also used in chemistry for chlorination and oxidation reactions.