Hydrogen bromide
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Hydrogen bromide?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Hydrogen bromide is the inorganic compound with the formula HBr. It is a hydrogen halide consisting of hydrogen and bromine. A colorless gas, it dissolves in water, forming hydrobromic acid, which is saturated at 68.85% HBr by weight at room temperature. Aqueous solutions that are 47.6% HBr by mass form a constant-boiling azeotrope mixture that boils at 124.3 °C (255.7 °F). Boiling less concentrated solutions releases H2O until the constant-boiling mixture composition is reached.
Quick Facts Names, Identifiers ...
 | |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Hydrogen bromide | |||
| Preferred IUPAC name
Bromane[1] | |||
| Identifiers | |||
3D model (JSmol) |
|||
| 3587158 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider |
| ||
| ECHA InfoCard | 100.030.090 | ||
| EC Number |
| ||
| KEGG |
| ||
| MeSH | Hydrobromic+Acid | ||
PubChem CID |
|||
| RTECS number |
| ||
| UNII | |||
| UN number | 1048 | ||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| HBr | |||
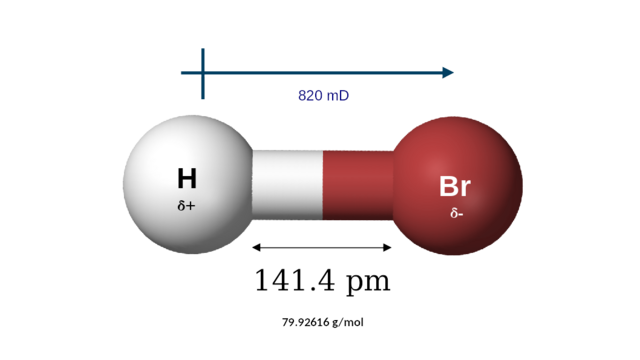
| Molar mass | 80.91 g/mol | ||
| Appearance | Colorless gas | ||
| Odor | Acrid | ||
| Density | 3.307 g/L (25 °C)[2] | ||
| Melting point | −86.9 °C (−124.4 °F; 186.2 K) | ||
| Boiling point | −66.8 °C (−88.2 °F; 206.3 K) | ||
| 221 g/100 mL (0 °C) 204 g/100 mL (15 °C) 193 g/100 mL (20 °C) 130 g/100 mL (100 °C) | |||
| Solubility | Soluble in alcohol, organic solvents | ||
| Vapor pressure | 2.308 MPa (at 21 °C) | ||
| Acidity (pKa) | −8.8 (±0.8);[3] ~−9[4] | ||
| Basicity (pKb) | ~23 | ||
| Conjugate acid | Bromonium | ||
| Conjugate base | Bromide | ||
Refractive index (nD) |
1.325[citation needed] | ||
| Structure | |||
| Linear | |||
| 820 mD | |||
| Thermochemistry | |||
Heat capacity (C) |
350.7 mJ/(K·g) | ||
Std molar entropy (S⦵298) |
198.696–198.704 J/(K·mol)[5] | ||
Std enthalpy of formation (ΔfH⦵298) |
−36.45...−36.13 kJ/mol[5] | ||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards |
Highly corrosive | ||
| GHS labelling: | |||
  | |||
| Danger | |||
| H314, H335 | |||
| P261, P280, P305+P351+P338, P310 | |||
| NFPA 704 (fire diamond) | |||
| Lethal dose or concentration (LD, LC): | |||
LC50 (median concentration) |
2858 ppm (rat, 1 h) 814 ppm (mouse, 1 h)[6] | ||
| NIOSH (US health exposure limits): | |||
PEL (Permissible) |
TWA 3 ppm (10 mg/m3)[7] | ||
REL (Recommended) |
TWA 3 ppm (10 mg/m3)[7] | ||
IDLH (Immediate danger) |
30 ppm[7] | ||
| Safety data sheet (SDS) | hazard.com | ||
| Related compounds | |||
Related compounds |
Hydrogen fluoride Hydrogen chloride Hydrogen iodide Hydrogen astatide | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Close
Hydrogen bromide, and its aqueous solution, hydrobromic acid, are commonly used reagents in the preparation of bromide compounds.