
Histone-like nucleoid-structuring protein
From Wikipedia, the free encyclopedia
Histone-like nucleoid-structuring protein (H-NS), is one of twelve nucleoid-associated proteins (NAPs)[1] whose main function is the organization of genetic material, including the regulation of gene expression via xenogeneic silencing.[2] H-NS is characterized by an N-terminal domain (NTD) consisting of two dimerization sites, a linker region that is unstructured and a C-terminal domain (CTD) that is responsible for DNA-binding.[2] Though it is a small protein (15 kDa),[3] it provides essential nucleoid compaction and regulation of genes (mainly silencing)[2] and is highly expressed, functioning as a dimer or multimer.[3] Change in temperature causes H-NS to be dissociated from the DNA duplex, allowing for transcription by RNA polymerase, and in specific regions lead to pathogenic cascades in enterobacteria such as Escherichia coli and the four Shigella species.[3]
| H-NS | |||||||||
|---|---|---|---|---|---|---|---|---|---|
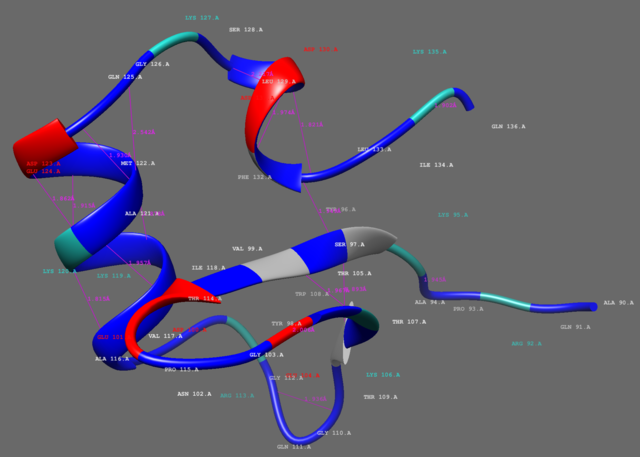
 Solution structure of the N-terminal domain (oligomerization domain) of the bacterial nucleoid structuring protein, H-NS. | |||||||||
| Identifiers | |||||||||
| Symbol | H-NS | ||||||||
| Pfam | PF00816 | ||||||||
| InterPro | IPR001801 | ||||||||
| CATH | [ P0ACF8] | ||||||||
| SCOP2 | 1hns / SCOPe / SUPFAM | ||||||||
| |||||||||