Loading AI tools
Chemical compound From Wikipedia, the free encyclopedia
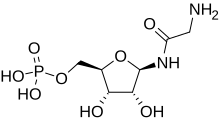
Glycineamide ribonucleotide (or GAR) is a biochemical intermediate in the formation of purine nucleotides via inosine-5-monophosphate, and hence is a building block for DNA and RNA.[1][2][3] The vitamins thiamine[4] and cobalamin[5] also contain fragments derived from GAR.[6]
 | |
| Names | |
|---|---|
| IUPAC name
(1R)-1,4-Anhydro-1-glycinamido-D-ribitol 5-(dihydrogen phosphate) | |
| Systematic IUPAC name
[(2R,3S,4R,5R)-5-(2-Aminoacetamido)-3,4-dihydroxyoxolan-2-yl]methyl dihydrogen phosphate | |
| Other names
Glycineamide ribotide, GAR | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| KEGG | |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C7H15N2O8P | |
| Molar mass | 286.177 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
GAR is the product of the enzyme phosphoribosylamine—glycine ligase acting on phosphoribosylamine (PRA) to combine it with glycine in a process driven by ATP. The reaction, EC 6.3.4.13 forms an amide bond:[7]
The biosynthesis pathway next adds a formyl group from 10-formyltetrahydrofolate to GAR, catalysed by phosphoribosylglycinamide formyltransferase in reaction EC 2.1.2.2 and producing formylglycinamide ribotide (FGAR):[7]
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.