
Galvanic cell
Electrochemical device / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Galvanic cell?
Summarize this article for a 10 year old
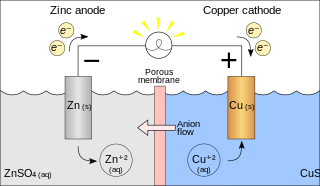
A galvanic cell or voltaic cell, named after the scientists Luigi Galvani and Alessandro Volta, respectively, is an electrochemical cell in which an electric current is generated from spontaneous oxidation–reduction reactions. A common apparatus generally consists of two different metals, each immersed in separate beakers containing their respective metal ions in solution that are connected by a salt bridge or separated by a porous membrane.[1]
Volta was the inventor of the voltaic pile, the first electrical battery. Common usage of the word battery has evolved to include a single Galvanic cell, but the first batteries had many Galvanic cells.[2]