
Favipiravir
Experimental antiviral drug with potential activity against RNA viruses / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Favipiravir?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
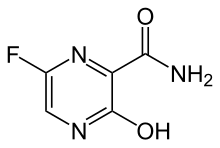
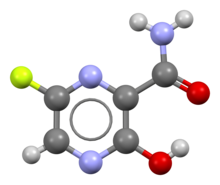
Favipiravir, sold under the brand name Avigan among others,[3] is an antiviral medication used to treat influenza in Japan.[4] It is also being studied to treat a number of other viral infections, including SARS-CoV-2.[4] Like the experimental antiviral drugs T-1105 and T-1106, it is a pyrazinecarboxamide derivative.[5]
You can help expand this article with text translated from the corresponding article in German. (March 2021) Click [show] for important translation instructions.
|
Quick Facts Clinical data, Trade names ...
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Avigan (アビガン, Abigan), Avifavir,[1] Areplivir,[2] others |
| Other names | T-705, favipira, favilavir |
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C5H4FN3O2 |
| Molar mass | 157.104 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Close
It is being developed and manufactured by Toyama Chemical (a subsidiary of Fujifilm) and was approved for medical use in Japan in 2014.[6] In 2016, Fujifilm licensed it to Zhejiang Hisun Pharmaceutical Co.[7] It became a generic drug in 2019.[citation needed]