
Erythrose
Chemical compound / From Wikipedia, the free encyclopedia
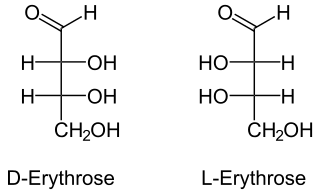
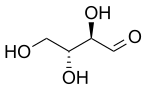
Erythrose is a tetrose saccharide with the chemical formula C4H8O4. It has one aldehyde group, and is thus part of the aldose family. The natural isomer is D-erythrose; it is a diastereomer of D-threose.[2]
Quick Facts Names, Identifiers ...
 D-Erythrose | |
 L-Erythrose | |
| Names | |
|---|---|
| IUPAC names
(2R,3R)-2,3,4-Trihydroxybutanal (D) (2S,3S)-2,3,4-Trihydroxybutanal (L) | |
| Identifiers | |
3D model (JSmol) |
|
| 5805561 | |
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.008.643 |
| EC Number |
|
| KEGG | |
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C4H8O4 | |
| Molar mass | 120.104 g·mol−1 |
| Appearance | Light yellow syrup |
| highly soluble | |
| Hazards | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
Erythrose was first isolated in 1849 from rhubarb by the French pharmacist Louis Feux Joseph Garot (1798-1869),[3] and was named as such because of its red hue in the presence of alkali metals (ἐρυθρός, "red").[4][5]
Erythrose 4-phosphate is an intermediate in the pentose phosphate pathway[6] and the Calvin cycle.[7]
Oxidative bacteria can be made to use erythrose as its sole energy source.[8]
