
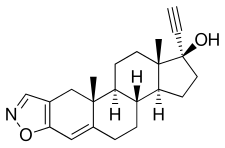
Danazol
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Danazol?
Summarize this article for a 10 year old
Danazol, sold as Danocrine and other brand names, is a medication used in the treatment of endometriosis, fibrocystic breast disease, hereditary angioedema and other conditions.[6][2][10][11][12] It is taken by mouth.[2]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Danatrol, Danocrine, Danol, Danoval, others |
| Other names | WIN-17757; 2,3-Isoxazolethisterone; 2,3-Isoxazol-17α-ethynyltestosterone; 17α-Ethynyl-17β-hydroxyandrost-4-en-[2,3-d]isoxazole |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a682599 |
| Pregnancy category |
|
| Routes of administration | By mouth |
| Drug class | Androgen; Anabolic steroid; Progestogen; Progestin; Antigonadotropin; Steroidogenesis inhibitor; Antiestrogen |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Saturable with dosage, higher with food intake[2] |
| Protein binding | To albumin, SHBGTooltip sex hormone-binding globulin, CBGTooltip corticosteroid-binding globulin[3][4][5] |
| Metabolism | Liver (CYP3A4)[6][7] |
| Metabolites | • 2-OHM-Ethisterone[7] • Ethisterone[8][9] |
| Elimination half-life | Acute: 3–10 hours[6][2] Chronic: 24–26 hours[6] |
| Excretion | Urine, feces[6][2] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.037.503 |
| Chemical and physical data | |
| Formula | C22H27NO2 |
| Molar mass | 337.463 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
The use of danazol is limited by masculinizing side effects such as acne, excessive hair growth, and voice deepening.[2][13] Danazol has a complex mechanism of action, and is characterized as a weak androgen and anabolic steroid, a weak progestogen, a weak antigonadotropin, a weak steroidogenesis inhibitor, and a functional antiestrogen.[5][14][15][16]
Danazol was discovered in 1963 and was introduced for medical use in 1971.[14][17][18][19] Due to their improved side-effect profiles, particularly their lack of masculinizing side effects, danazol has largely been replaced by gonadotropin-releasing hormone analogues (GnRH analogues) in the treatment of endometriosis.[4]