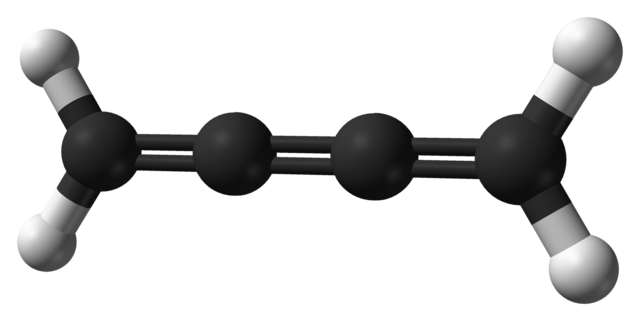
Cumulene
Hydrocarbon compound with ≥3 consecutive double bonds / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Cumulene?
Summarize this article for a 10 year old
A cumulene is a compound having three or more cumulative (consecutive) double bonds.[1] They are analogous to allenes, only having a more extensive chain. The simplest molecule in this class is butatriene (H2C=C=C=CH2), which is also called simply cumulene. Unlike most alkanes and alkenes, cumulenes tend to be rigid, comparable to polyynes. Cumulene carbenes H2Cn for n from 3 to 6 have been observed in interstellar molecular clouds[2][3] and in laboratory experiments[4] by using microwave and infrared spectroscopy. (The more stable cumulenes H2CnH2 are difficult to detect optically because they lack an electric dipole moment.) Cumulenes containing heteroatoms are called heterocumulenes;[5] an example is carbon suboxide.