
Clobazam
Benzodiazepine class medication / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Clobazam?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
"Onfi" redirects here. For the ONFI standards consortium, see Open NAND Flash Interface Working Group.
Clobazam, sold under the brand names Frisium, Onfi and others, is a benzodiazepine class medication that was patented in 1968.[3] Clobazam was first synthesized in 1966 and first published in 1969. Clobazam was originally marketed as an anxioselective anxiolytic since 1970,[4][5] and an anticonvulsant since 1984.[6] The primary drug-development goal was to provide greater anxiolytic, anti-obsessive efficacy with fewer benzodiazepine-related side effects.[4]
Quick Facts Clinical data, Trade names ...
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Frisium, Urbanol, Onfi, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a612008 |
| License data | |
| Pregnancy category |
|
| Routes of administration | By mouth |
| Drug class | Benzodiazepine |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 87% (oral) |
| Protein binding | 80–90% |
| Metabolism | Liver |
| Metabolites |
|
| Onset of action | 0.5–4 hours |
| Elimination half-life |
|
| Excretion | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.040.810 |
| Chemical and physical data | |
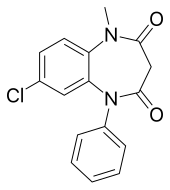
| Formula | C16H13ClN2O2 |
| Molar mass | 300.74 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Close