
Caesium oxalate
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Caesium oxalate?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
Not to be confused with cerium oxalate.
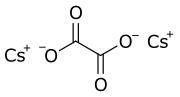
Caesium oxalate (standard IUPAC spelling), or dicesium oxalate, or cesium oxalate (American spelling) is a chemical compound with the chemical formula Cs2C2O4. It is a caesium salt of oxalic acid. It consists of caesium cations Cs+ and oxalate anions C2O2−4.
Quick Facts Names, Identifiers ...
 | |
| Names | |
|---|---|
| IUPAC name
Caesium oxalate | |
| Preferred IUPAC name
Dicaesium oxalate | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.012.683 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| Cs2C2O4 | |
| Molar mass | 353.829 g·mol−1 |
| Appearance | White solid |
| 76 g/(100 ml) (25 °C)[1] | |
| Hazards | |
| GHS labelling: | |
 | |
| Warning | |
| H302, H312 | |
| P264, P270, P280, P301+P312, P302+P352, P312, P322, P330, P363, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close