
Aminopterin
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Aminopterin?
Summarize this article for a 10 year old
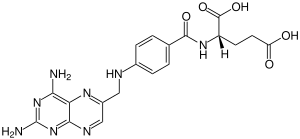
Aminopterin (or 4-aminopteroic acid), the 4–amino derivative of folic acid, is an antineoplastic drug with immunosuppressive properties often used in chemotherapy. Aminopterin is a synthetic derivative of pterin. Aminopterin works as an enzyme inhibitor by competing for the folate binding site of the enzyme dihydrofolate reductase. Its binding affinity for dihydrofolate reductase effectively blocks tetrahydrofolate synthesis. This results in the depletion of nucleotide precursors and inhibition of DNA, RNA, and protein synthesis.
 | |
 | |
| Names | |
|---|---|
| IUPAC name
N-(4-{[(2,4-Diaminopteridin-6-yl)methyl]amino}benzoyl)-L-glutamic acid | |
| Systematic IUPAC name
(2S)-2-(4-{[(2,4-Diaminopteridin-6-yl)methyl]amino}benzamido)pentanedioic acid | |
| Other names
4-Aminofolic acid 4-Aminopteroylglutamic acid Aminopterin sodium Aminopteroylglutamic acid | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | 100.000.191 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C19H20N8O5 | |
| Molar mass | 440.41 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
It is classified as an extremely hazardous substance in the United States as defined in Section 302 of the U.S. Emergency Planning and Community Right-to-Know Act (42 U.S.C. 11002), and is subject to strict reporting requirements by facilities which produce, store, or use it in significant quantities.[1]