
2C (psychedelics)
Family of phenethylamine psychedelics / From Wikipedia, the free encyclopedia
2C (2C-x) is a general name for the family of psychedelic phenethylamines containing methoxy groups on the 2 and 5 positions of a benzene ring.[1] Most of these compounds also carry lipophilic substituents at the 4 position, usually resulting in more potent and more metabolically stable and longer acting compounds.[2] Most of the currently known 2C compounds were first synthesized by Alexander Shulgin in the 1970s and 1980s and published in his book PiHKAL (Phenethylamines i Have Known And Loved). Shulgin also coined the term 2C, being an acronym for the 2 carbon atoms between the benzene ring and the amino group.[3]

More information Nomenclature, R3 ...
| Nomenclature | R3 | R4 | 2D Structure | CAS number |
|---|---|---|---|---|
| 2C-B | H | Br |  |
66142-81-2 |
| 2C-Bn | H | CH2C6H5 | 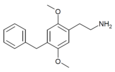 |
|
| 2C-Bu | H | CH2CH2CH2CH3 |  |
|
| 2C-C | H | Cl |  |
88441-14-9 |
| 2C-C-3 [4] | Cl | Cl |  |
|
| 2C-CN | H | C≡N | 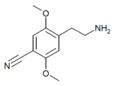 |
88441-07-0 |
| 2C-D | H | CH3 | 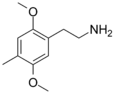 |
24333-19-5 |
| 2C-E | H | CH2CH3 |  |
71539-34-9 |
| 2C-EF | H | CH2CH2F |  |
1222814-77-8 |
| 2C-F | H | F | 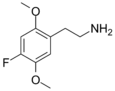 |
207740-15-6 |
| 2C-G | CH3 | CH3 | 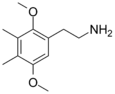 |
207740-18-9 |
| 2C-G-1 | CH2 |  |
||
| 2C-G-2 | (CH2)2 | 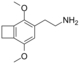 |
||
| 2C-G-3 | (CH2)3 |  |
207740-19-0 | |
| 2C-G-4 | (CH2)4 |  |
952006-59-6 | |
| 2C-G-5 | (CH2)5 |  |
207740-20-3 | |
| 2C-G-6 | (CH2)6 |  |
||
| 2C-G-N | (CH)4 |  |
207740-21-4 | |
| 2C-H | H | H | 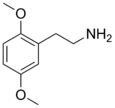 |
3600-86-0 |
| 2C-I | H | I | 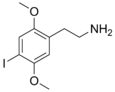 |
69587-11-7 |
| 2C-iP | H | CH(CH3)2 |  |
1498978-47-4 |
| 2C-TBU | H | C(CH3)3 | 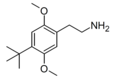 |
|
| 2C-CP | H | C3H5 |  |
2888537-46-8 |
| 2C-CPE | H | C5H9 |  |
|
| 2C-N | H | NO2 |  |
261789-00-8 |
| 2C-NH2 | H | NH2 |  |
168699-66-9 |
| 2C-PYR | H | Pyrrolidine |  |
|
| 2C-PIP | H | Piperidine | 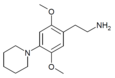 |
|
| 2C-O | H | OCH3 | 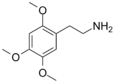 |
15394-83-9 |
| 2C-O-4 | H | OCH(CH3)2 | 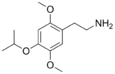 |
952006-65-4 |
| 2C-MOM [5] | H | CH2OCH3 |  |
|
| 2C-P | H | CH2CH2CH3 |  |
207740-22-5 |
| 2C-Ph | H | C6H5 |  |
|
| 2C-Se | H | Se CH3 |  |
1189246-68-1 |
| 2C-T | H | SCH3 | 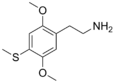 |
61638-09-3 |
| 2C-T-2 | H | SCH2CH3 | 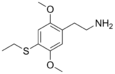 |
207740-24-7 |
| 2C-T-3[6] | H | SCH2C(=CH2)CH3 |  |
648957-40-8 |
| 2C-T-4 | H | SCH(CH3)2 | 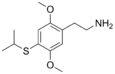 |
207740-25-8 |
| 2C-T-5[6] |  |
|||
| 2C-T-6[6] | 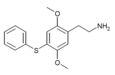 |
|||
| 2C-T-7 | H | S(CH2)2CH3 | 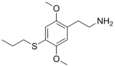 |
207740-26-9 |
| 2C-T-8 | H | SCH2CH(CH2)2 | 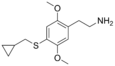 |
207740-27-0 |
| 2C-T-9[6] |  |
207740-28-1 | ||
| 2C-T-10[6] |  |
|||
| 2C-T-11[6] | 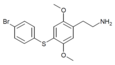 |
|||
| 2C-T-12[6] |  |
|||
| 2C-T-13 | H | S(CH2)2OCH3 |  |
207740-30-5 |
| 2C-T-14[6] | 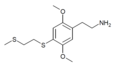 |
|||
| 2C-T-15 | H | SCH(CH2)2 |  |
|
| 2C-T-16[7] | H | SCH2CH=CH2 |  |
648957-42-0 |
| 2C-T-17 | H | SCH(CH3)CH2CH3 | 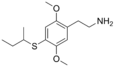 |
207740-32-7 |
| 2C-T-18[6] |  |
|||
| 2C-T-19 | H | SCH2CH2CH2CH3 | 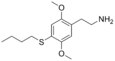 |
|
| 2C-T-21 | H | S(CH2)2F |  |
207740-33-8 |
| 2C-T-21.5[6] |  |
648957-46-4 | ||
| 2C-T-22[6] |  |
648957-48-6 | ||
| 2C-T-23[6] | 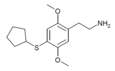 |
|||
| 2C-T-24[6] |  |
|||
| 2C-T-25[6] |  |
|||
| 2C-T-27[6] |  |
648957-52-2 | ||
| 2C-T-28[6] |  |
648957-54-4 | ||
| 2C-T-30[6] | 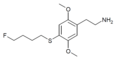 |
|||
| 2C-T-31[6] | 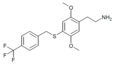 |
|||
| 2C-T-32[6] |  |
|||
| 2C-T-33[6] |  |
|||
| 2C-T-DFM | H | SCF2H |  |
|
| CYB210010[8] | H | SCF3 | 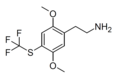 |
|
| 2C-T-DFP | H | SCH2CH2CF2H |  |
|
| 2C-T-PARGY | H | SCH2C≡CH | 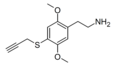 |
|
| 2C-DFM [9]: 770 | H | CHF2 | 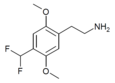 |
|
| 2C-TFM | H | CF3 |  |
159277-08-4 |
| 2C-TFE | H | CH2CF3 | 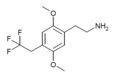 |
|
| 2C-PFE | H | CF2CF3 |  |
|
| 2C-PFS | H | SF5 | 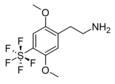 |
|
| 2C-YN | H | C≡CH | 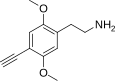 |
752982-24-4 |
| 2C-V | H | CH=CH2 | 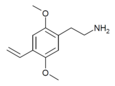 |
|
| 2C-AL[10] | H | CH2CH=CH2 | 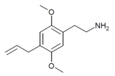 |
|
Close