
2,3-Dimethylpentane
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about 2,3-Dimethylpentane?
Summarize this article for a 10 year old
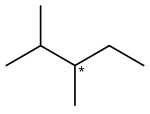
2,3-Dimethylpentane is an organic compound of carbon and hydrogen with formula C
7H
16, more precisely CH
3–CH(CH
3)–CH(CH
3)–CH
2–CH
3: a molecule of pentane with methyl groups –CH
3 replacing hydrogen atoms on carbon atoms 2 and 3. It is an alkane ("paraffin" in older nomenclature), a fully saturated hydrocarbon; specifically, one of the isomers of heptane.
 | |
| Names | |
|---|---|
| Preferred IUPAC name
2,3-Dimethylpentane | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI |
|
| ChemSpider | |
| ECHA InfoCard | 100.008.437 |
| EC Number |
|
PubChem CID |
|
| UNII |
|
| UN number | 1206 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C7H16 | |
| Molar mass | 100.205 g·mol−1 |
| Appearance | Colourless liquid |
| Density | 0.7076 g/mL (25 °C), 0.6413 (80 °C), 0.7380 (25 °C, 45 MPa), 0.6891 (80 °C, 45 MPa) (racemic)[1] |
| Boiling point | 89.7 °C (racemic)[2][3][4] |
| Vapor pressure | 2.35 psi (37.7 °C)[5] |
| Viscosity | 0.356 mPa s (30 °C), 0.232 (80 °C), 0.624 (30 °C, 60 MPa) (racemic)[1] |
| Thermochemistry | |
Heat capacity (C) |
34.308 cal/K/mol (−189 °C), 51.647 (20 °C), 58.735 (86.6 °C) (racemic)[6] |
Std molar entropy (S⦵298) |
71.02 cal/K/mol (25 °C) (racemic)[6] |
| Hazards | |
| GHS labelling: | |
    | |
| Danger | |
| H225, H304, H315, H335, H336, H410 | |
| P210, P233, P240, P241, P242, P243, P261, P264, P271, P273, P280, P301+P310, P302+P352, P303+P361+P353, P304+P340, P312, P321, P331, P332+P313, P362, P370+P378, P391, P403+P233, P403+P235, P405, P501 | |
| Flash point | −7 °C (19 °F; 266 K)[5] |
| 337 °C (639 °F; 610 K)[5] | |
| Related compounds | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Like typical alkanes, it is a colorless flammable compound; under common ambient conditions, it is a mobile liquid, less dense than water.[1]
2,3-Dimethylpentane is notable for being one of the two simplest alkanes with optical (enantiomeric) isomerism. The optical center is the middle carbon of the pentane backbone, which is connected to one hydrogen atom, one methyl group, one ethyl group –C
2H
5, and one isopropyl group –CH(CH
3)
2. The two enantiomers are denoted (3R)-2,3-dimethylpentane and (3S)-2,3-dimethylpentane. (The other simplest chiral alkane is its structural isomer 3-methylhexane.)