
1,4-Dihydroxyanthraquinone
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about 1,4-Dihydroxyanthraquinone?
Summarize this article for a 10 year old
SHOW ALL QUESTIONS
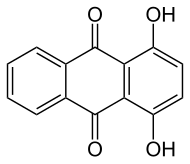
1,4-Dihydroxyanthraquinone, also called quinizarin or Solvent Orange 86, is an organic compound derived from anthroquinone. Quinizarin is an orange or red-brown crystalline powder. It is formally derived from anthraquinone by replacement of two hydrogen atoms by hydroxyl (OH) groups. It is one of ten dihydroxyanthraquinone isomers and occurs in small amounts (as a glycoside) in the root of the madder plant, Rubia tinctorum.[1]
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
1,4-Dihydroxyanthracene-9,10-dione | |
| Other names
Quinizarin; Solvent Orange 86 | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider |
|
| ECHA InfoCard | 100.001.245 |
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C14H8O4 | |
| Molar mass | 240.21 g/mol |
| Appearance | Orange or red-brown crystalline powder |
| Melting point | 198 to 199 °C (388 to 390 °F; 471 to 472 K) |
| Boiling point | 450 °C (842 °F; 723 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close