1,2-Dithiolane
Chemical compound / From Wikipedia, the free encyclopedia
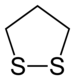
1,2-Dithiolane is an organosulfur compound with the formula S2(CH2)3. It is also classified as a heterocycle derived from cyclopentane by replacing two methylene bridges (-CH
2- units) with a disulfide group. 1,3-Dithiolane is an isomer. The parent molecule is not important but substituted derivatives, especially lipoic acid and its derivatives, are important. Several occur in foods.[1]
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
1,2-Dithiolane | |||
| Identifiers | |||
| |||
3D model (JSmol) |
|||
| 102454 | |||
| ChEBI | |||
| ChemSpider | |||
| 1029938 | |||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| C3H6S2 | |||
| Molar mass | 106.20 g·mol−1 | ||
| Related compounds | |||
Related compounds |
Ethane-1,2-dithiol | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
1,2-Dithiolane is the disulfide derived from 1,3-propanedithiol. It is however unstable with respect to polymerization.[2]
Substituted derivatives of 1,2-dithiolane are found in some foods, such as asparagusic acid in asparagus.[3] The 4-dimethylamino derivative nereistoxin was the inspiration for insecticides that act by blocking the nicotinic acetylcholine receptor.[4]
Lipoic acid is essential for aerobic metabolism in mammals and also has strong affinity with many metals including gold, molybdenum, and tungsten.[5] Other 1,2-dithiolanes have relevance in nanomaterials such as gold nanoparticles or transition metal dichalcogenide monolayers (TMDs) (MoS2 and WS2).[6][7][8]
- asparagusic acid
- nereistoxin, from which insecticides including cartap and bensultap were derived
- lipoic acid