ስዕል:Sodium_chloride_crystal.png
From Wikipedia, the free encyclopedia
Sodium_chloride_crystal.png (410 × 372 ፒክስል፤ መጠን፦ 52 KB፤ የMIME ዓይነት፦ image/png)
ይህ ፋይል ከWikimedia Commons የተቀሰመ ነው እና በሌላ ዊኪ ላይ ሊጠቅም ይችላል። በhttps://commons.wikimedia.org/wiki/File:Sodium_chloride_crystal.png ላይ የሚገኘው የፋይሉ መግለጫ ከዚህ በታች ቀርቧል።

|
File:Ionlattice-fcc.svg is a vector version of this file. It should be used in place of this PNG file when not inferior.
File:Sodium chloride crystal.png → File:Ionlattice-fcc.svg
For more information, see Help:SVG.
|
| ማጠቃለያSodium chloride crystal.png |
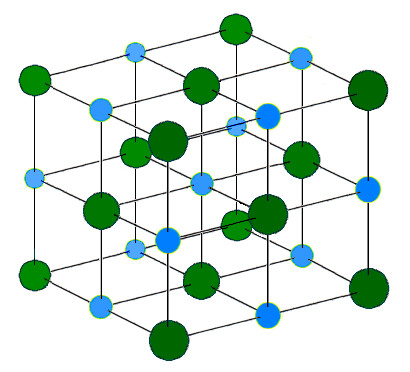
The Sodium Chloride Crystal Structure. Each atom has six nearest neighbors, with octahedral geometry. This arrangement is known as cubic close packed (ccp) or face-centered cubic (fcc). Light blue = Na+ |
| ቀን | UTC |
| ምንጭ | H Padleckas made this image file by June 2005 for use in the articles "Sodium Chloride" and "Ionic Crystal" in Wikipedia by modifying the existing image file en:Image:Sodium_chloride_crystal.jpg from Wikipedia created and released into the public domain by User:Walkerma. |
| አቅራቢው | H Padleckas |
| ሌሎች ዕትሞች | en:Image:Sodium_chloride_crystal.jpg |
| Public domainPublic domainfalsefalse |
| I, the copyright holder of this work, release this work into the public domain. This applies worldwide. In some countries this may not be legally possible; if so: I grant anyone the right to use this work for any purpose, without any conditions, unless such conditions are required by law. |
Captions
Add a one-line explanation of what this file represents
Steenzout kristalstructuur
Items portrayed in this file
depicts እንግሊዝኛ
creator እንግሊዝኛ
some value
media type እንግሊዝኛ
image/png
የፋይሉ ታሪክ
የቀድሞው ዕትም ካለ ቀን/ሰዓቱን በመጫን መመልከት ይቻላል።
| ቀን /ሰዓት | ናሙና | ክልሉ (በpixel) | አቅራቢው | ማጠቃለያ | |
|---|---|---|---|---|---|
| ያሁኑኑ | 23:27, 9 ፌብሩዌሪ 2023 |  | 410 × 372 (52 KB) | Materialscientist | transparent background |
| 23:54, 14 ጁን 2005 |  | 410 × 372 (37 KB) | H Padleckas | The Sodium Chloride Crystal Structure. Each atom has six nearest neighbors, with octahedral geometry. This arrangement is known as ''cubic close packed'' (ccp). Light blue = Na<sup>+</sup> Dark green = Cl<sup>-</sup> |
መያያዣዎች
የሚከተለው ገጽ ወደዚሁ ፋይል ተያይዟል፦
ሌሎች ውኪዎች
የሚከተሉት ሌሎች ውኪዎች ይህን ፋይል ይጠቀማሉ፦
- Usage on ar.wikipedia.org
- Usage on bs.wikipedia.org
- Usage on ca.wikipedia.org
- Usage on co.wikipedia.org
- Usage on cs.wikipedia.org
- Usage on de.wikipedia.org
- Usage on de.wikibooks.org
- Usage on el.wikipedia.org
- Usage on el.wikibooks.org
- Usage on en.wikipedia.org
- Usage on en.wikibooks.org
- Organic Chemistry/Foundational concepts of organic chemistry/Bonding
- General Chemistry/Introduction
- General Chemistry/Ionic bonding
- Organic Chemistry/Print version
- Adventist Youth Honors Answer Book/Nature/Rocks & Minerals - Advanced
- General Chemistry/Print version
- User:Jplego/Collections
- User:Jplego/Gen Chem
- Usage on en.wikiversity.org
- Usage on eo.wikipedia.org
- Usage on es.wikipedia.org
- Usage on fi.wikipedia.org
- Usage on fr.wikipedia.org
- Usage on fr.wikiversity.org
- Usage on fr.wiktionary.org
- Usage on ga.wikipedia.org
- Usage on he.wikipedia.org
- Usage on hr.wikipedia.org
- Usage on hu.wikipedia.org
- Usage on id.wikibooks.org
- Usage on it.wikipedia.org
- Usage on ko.wikipedia.org
- Usage on la.wikipedia.org
- Usage on lt.wikipedia.org
- Usage on mk.wikipedia.org
View more global usage of this file.
ተጨማሪ መረጃ
ይህ ፋይል በውስጡ ተጨማሪ መረጃ ይይዛል። መረጃውም በዲጂታል ካሜራ ወይም በኮምፒውተር ስካነር የተጨመረ ይሆናል። ይህ ከኦሪጂናሉ ቅጅ የተለወጠ ከሆነ፣ ምናልባት የመረጃው ዝርዝር ለውጦቹን የማያንጸባረቅ ይሆናል።
| አድማሳዊ ማጉላት | 157.46 dpc |
|---|---|
| ቁም ማጉላት | 157.46 dpc |
| ፋይሉ የተቀየረበት ቀንና ሰዓት | 23:32, 9 ፌብሩዌሪ 2023 |