
Water splitting
Chemical reaction / From Wikipedia, the free encyclopedia
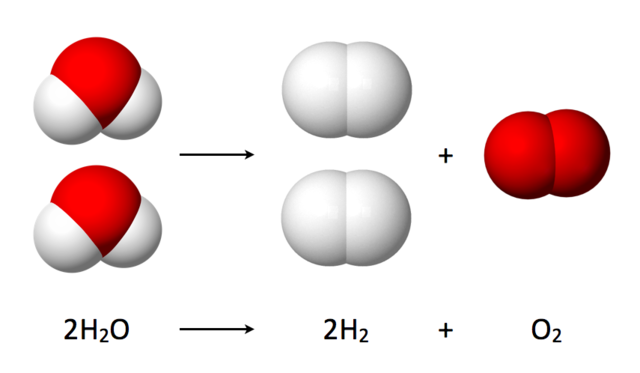
Water splitting is the chemical reaction in which water is broken down into oxygen and hydrogen:[1]
2 H2O → 2 H2 + O2
Efficient and economical water splitting would be a technological breakthrough that could underpin a hydrogen economy. A version of water splitting occurs in photosynthesis, but hydrogen is not produced. The reverse of water splitting is the basis of the hydrogen fuel cell. Water splitting using solar radiation has not been commercialized.