Strecker amino acid synthesis
Method for the synthesis of amino acids / From Wikipedia, the free encyclopedia
The Strecker amino acid synthesis, also known simply as the Strecker synthesis, is a method for the synthesis of amino acids by the reaction of an aldehyde with cyanide in the presence of ammonia. The condensation reaction yields an α-aminonitrile, which is subsequently hydrolyzed to give the desired amino acid.[1][2] The method is used for the commercial production of racemic methionine from methional.[3]
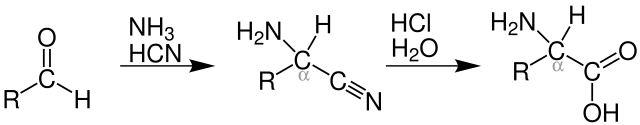
Quick Facts Identifiers ...
| Strecker synthesis | |
|---|---|
| Named after | Adolph Strecker |
| Reaction type | Substitution reaction |
| Identifiers | |
| Organic Chemistry Portal | strecker-synthesis |
| RSC ontology ID | RXNO:0000207 |
Close
Primary and secondary amines also give N-substituted amino acids. Likewise, the usage of ketones, instead of aldehydes, gives α,α-disubstituted amino acids.[4]