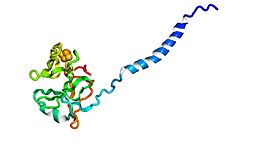
Rieske proteins are iron–sulfur protein (ISP) components of cytochrome bc1 complexes and cytochrome b6f complexes and are responsible for electron transfer in some biological systems. John S. Rieske and co-workers first discovered the protein and in 1964 isolated an acetylated form of the bovine mitochondrial protein.[1] In 1979, Trumpower's team isolated the "oxidation factor" from bovine mitochondria and showed it was a reconstitutively-active form of the Rieske iron-sulfur protein [2]
It is a unique [2Fe-2S] cluster in that one of the two Fe atoms is coordinated by two histidine residues rather than two cysteine residues. They have since been found in plants, animals, and bacteria with widely ranging electron reduction potentials from -150 to +400 mV.[3]
Quick Facts Identifiers, Symbol ...
Close
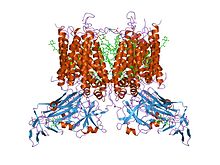
Quick Facts Cytochrome B6-F complex Fe-S subunit, alpha helical transmembrane domain, Identifiers ...
| Cytochrome B6-F complex Fe-S subunit, alpha helical transmembrane domain |
|---|
crystal structure of cytochrome b6f complex from m.laminosus |
|
| Symbol | CytB6-F_Fe-S |
|---|
| Pfam | PF08802 |
|---|
| InterPro | IPR014909 |
|---|
|
Close