
Precipitation (chemistry)
Chemical process leading to the settling of an insoluble solid from a solution / From Wikipedia, the free encyclopedia
This article is about the chemical phenomenon. For other uses, see Precipitation (disambiguation).
"Precipitate" redirects here. For other uses, see Precipitate (disambiguation).
In an aqueous solution, precipitation is the "sedimentation of a solid material (a precipitate) from a liquid solution".[1][2] The solid formed is called the precipitate.[3] In case of an inorganic chemical reaction leading to precipitation, the chemical reagent causing the solid to form is called the precipitant.[4]
This article has multiple issues. Please help improve it or discuss these issues on the talk page. (Learn how and when to remove these template messages)
|
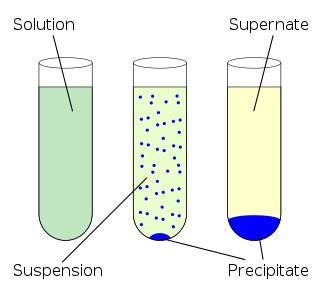
The clear liquid remaining above the precipitated or the centrifuged solid phase is also called the supernate or supernatant.
The notion of precipitation can also be extended to other domains of chemistry (organic chemistry and biochemistry) and even be applied to the solid phases (e.g. metallurgy and alloys) when solid impurities segregate from a solid phase.