
Phosphorus heptabromide
Chemical compound / From Wikipedia, the free encyclopedia
For the pigment code PBr7, see List of inorganic pigments § Brown pigments.
Phosphorus heptabromide is an inorganic compound with the chemical formula PBr7. It is one of the phosphorus bromides. At normal conditions, it forms red prismatic crystals. PBr7 can be prepared by the sublimation of a mixture of phosphorus pentabromide and bromine.[2]
- PBr5 + Br2 → PBr7
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Tetrabromophosphanium tribromide | |
Other names
| |
| Identifiers | |
3D model (JSmol) |
|
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| PBr7 | |
| Molar mass | 590.302 g·mol−1 |
| Appearance | Red prismatic crystals |
| Structure[1] | |
| Orthorhombic | |
| Pnma, No. 64 | |
a = 9.35 Å, b = 7.94 Å, c = 14.69 Å | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
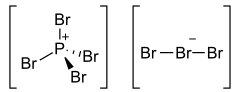
The structure of PBr7 consists of a tetrabromophosphonium cation [PBr4]+, paired with a tribromide anion [Br3]−, and the tribromide anion is non-symmetric.[1]