
Phosphonate
Organic compound containing C–PO(OR)2 groups / From Wikipedia, the free encyclopedia
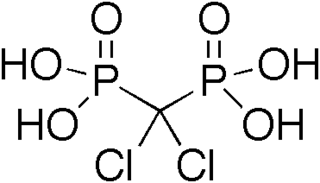
In organic chemistry, phosphonates or phosphonic acids are organophosphorus compounds containing C−PO(OR)2 groups, where R is an organic group (alkyl, aryl). If R is hydrogen then the compound is a dialkyl phosphite, which is a different functional group. Phosphonic acids, typically handled as salts, are generally nonvolatile solids that are poorly soluble in organic solvents, but soluble in water and common alcohols.

Many commercially important compounds are phosphonates, including glyphosate (the active molecule of the herbicide Roundup), and ethephon, a widely used plant growth regulator. Bisphosphonates are popular drugs for treatment of osteoporosis.[1]
In biochemistry and medicinal chemistry, phosphonate groups are used as stable bioisosteres for phosphate, such as in the antiviral nucleotide analog, Tenofovir, one of the cornerstones of anti-HIV therapy. And there is an indication that phosphonate derivatives are "promising ligands for nuclear medicine."[2]