
Neostigmine
Anti-full body paralysis drug treatment / From Wikipedia, the free encyclopedia
Neostigmine, sold under the brand name Bloxiverz, among others, is a medication used to treat myasthenia gravis, Ogilvie syndrome, and urinary retention without the presence of a blockage.[3][4] It is also used in anaesthesia to end the effects of non-depolarising neuromuscular blocking medication.[3] It is given by injection either into a vein, muscle, or under the skin.[3] After injection effects are generally greatest within 30 minutes and last up to 4 hours.[3][5]
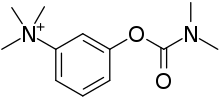 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Bloxiverz, Prostigmin, Vagostigmin, others |
| AHFS/Drugs.com | Monograph |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Intramuscular, intravenous, subcutaneous, by mouth |
| Drug class | Cholinesterase inhibitor |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | Unclear, probably less than 5% |
| Metabolism | Slow hydrolysis by acetylcholinesterase and also by plasma esterases |
| Onset of action | Within 10-20 min (injection),[3] with 4 hrs (by mouth) [citation needed] |
| Elimination half-life | 50–90 minutes |
| Duration of action | up to 4 hrs[3] |
| Excretion | Unchanged drug (up to 70%) and alcoholic metabolite (30%) are excreted in the urine |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.305.602 |
| Chemical and physical data | |
| Formula | C12H19N2O2+ |
| Molar mass | 223.296 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Common side effects include nausea, increased saliva, crampy abdominal pain, and slow heart rate.[3] More severe side effects include low blood pressure, weakness, and allergic reactions.[3] It is unclear if use in pregnancy is safe for the baby.[3] Neostigmine is in the cholinergic family of medications.[3] It works by blocking the action of acetylcholinesterase and therefore increases the levels of acetylcholine.[3]
Neostigmine was patented in 1931.[6] It is on the World Health Organization's List of Essential Medicines.[7] The term is from Greek neos, meaning "new", and "-stigmine", in reference to its parent molecule, physostigmine, on which it is based.[8] It is available as a generic medication.[9]