
Kelvin
SI unit of temperature / From Wikipedia, the free encyclopedia
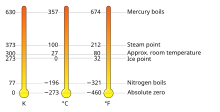
The kelvin, symbol K, is the base unit of measurement for temperature in the International System of Units (SI). The Kelvin scale is an absolute temperature scale that starts from 0 K, the lowest possible temperature (absolute zero), then rises by exactly 1 K for each 1 °C.[1][2][3][4] The Kelvin scale was designed to be easily converted from the Celsius scale (symbol °C). Any temperature in degrees Celsius can be converted to kelvin by adding 273.15.[1][5]
| kelvin | |
|---|---|
 | |
| General information | |
| Unit system | SI |
| Unit of | temperature |
| Symbol | K |
| Named after | William Thomson, 1st Baron Kelvin |
| 2019 definition | kB ≝ 1.380649×10−23 J/K |
| Conversions | |
| x K in ... | ... corresponds to ... |
| Celsius | (x − 273.15) °C |
| Fahrenheit | (1.8 x − 459.67) °F |
| Rankine | 1.8 x °Ra |
The 19th century British scientist Lord Kelvin first developed and proposed the scale.[5] It was often called the "absolute Celsius" scale in the early 20th century.[6] The kelvin was formally added to the International System of Units in 1954, defining 273.16 K to be the triple point of water. The Celsius, Fahrenheit, and Rankine scales were redefined in terms of the Kelvin scale using this definition.[2][7][8] The 2019 redefinition of the SI base units now defines the kelvin in terms of energy by setting the Boltzmann constant to exactly 1.380649×10−23 joules per kelvin;[2] every 1 K change of thermodynamic temperature corresponds to a thermal energy change of exactly 1.380649×10−23 J.