IMes
Chemical compound / From Wikipedia, the free encyclopedia
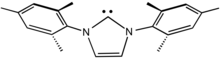
IMes is an abbreviation for an organic compound that is a common ligand in organometallic chemistry. It is an N-heterocyclic carbene (NHC). The compound, a white solid, is often not isolated but instead is generated upon attachment to the metal centre.[1]
Quick Facts Names, Identifiers ...
 | |
| Names | |
|---|---|
| Preferred IUPAC name
1,3-Bis(2,4,6-trimethylphenyl)-1,3-dihydro-2H-imidazol-2-ylidene | |
| Other names
1,3-Dimesitylimidazol-2-ylidene, 1,3-bis(2,4,6-trimethylphenyl)-imidazolium, 1,3-bis(2,4,6-trimethylphenyl)imidazol-2-ylidene | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.154.201 |
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C21H24N2 | |
| Molar mass | 304.43 |
| Appearance | white solid |
| Melting point | 150 to 155 °C (302 to 311 °F; 423 to 428 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
First prepared by Arduengo,[2] the heterocycle is synthesized by condensation of 2,4,6-trimethylaniline and glyoxal to give the diimine. In the presence of acid, the resulting glyoxal-bis(mesitylimine) condenses with formaldehyde to give the dimesitylimidazolium cation. This cation is the conjugate acid of the NHC.[3][4]