
Glyceraldehyde
Chemical compound / From Wikipedia, the free encyclopedia
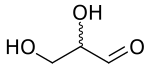
Glyceraldehyde (glyceral) is a triose monosaccharide with chemical formula C3H6O3. It is the simplest of all common aldoses. It is a sweet, colorless, crystalline solid that is an intermediate compound in carbohydrate metabolism. The word comes from combining glycerol and aldehyde, as glyceraldehyde is glycerol with one alcohol group oxidized to an aldehyde.
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Glyceraldehyde | |
| Systematic IUPAC name
2,3-Dihydroxypropanal | |
| Other names
Glyceraldehyde Glyceric aldehyde Glyceral | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.000.264 |
PubChem CID |
|
| UNII |
|
CompTox Dashboard (EPA) |
|
| |
| |
| Properties[1] | |
| C3H6O3 | |
| Molar mass | 90.078 g·mol−1 |
| Density | 1.455 g/cm3 |
| Melting point | 145 °C (293 °F; 418 K) |
| Boiling point | 140 to 150 °C (284 to 302 °F; 413 to 423 K) at 0.8 mmHg |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close