
Ferrichrome
Chemical compound / From Wikipedia, the free encyclopedia
Not to be confused with ferrochrome.
For the audio tape formulation, see Compact Cassette tape types and formulations § Ferrichrome.
Ferrichrome is a cyclic hexa-peptide that forms a complex with iron atoms. It is a siderophore composed of three glycine and three modified ornithine residues with hydroxamate groups [-N(OH)C(=O)C-]. The 6 oxygen atoms from the three hydroxamate groups bind Fe(III) in near perfect octahedral coordination.
Quick Facts Names, Identifiers ...
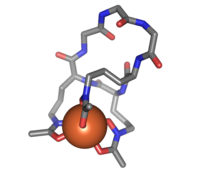 Ferrichrome (sticks) bound to an iron atom (orange) | |
| Names | |
|---|---|
| IUPAC name
N-[3-[4,16-bis[3-[acetyl(oxido)amino]propyl]-2,5,8,11,14,17-hexaoxo-3,6,9,12,15,18-hexazacyclooctadec-1-yl]propyl]-N-oxidoacetamide; iron(3+) | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.036.081 |
| EC Number |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C27H42FeN9O12 | |
| Molar mass | 740.529 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
Ferrichrome was first isolated in 1952, and has been found to be produced by fungi of the genera Aspergillus, Ustilago, and Penicillium.[1] However, at the time there was no understanding regarding its involvement and contribution to iron transport.[2] It was not until 1957 because of Joe Neilands' work, where he first noted that Ferrichrome was able to act as an iron transport agent.