
Drospirenone
Medication drug / From Wikipedia, the free encyclopedia
Drospirenone is a progestin and antiandrogen medication which is used in birth control pills to prevent pregnancy and in menopausal hormone therapy, among other uses.[1][9] It is available both alone under the brand name Slynd and in combination with an estrogen under the brand name Yasmin among others.[9][3] The medication is an analog of the drug spironolactone.[10] Drospirenone is taken by mouth.[1][3]
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation | Droe-SPY-re-nown |
| Trade names | Alone: Slynd With estradiol: Angeliq With ethinylestradiol: Yasmin, Yasminelle, Yaz, others With estetrol: Nextstellis |
| Other names | Dihydrospirenone; Dihydrospirorenone; 1,2-Dihydrospirorenone; MSp; SH-470; ZK-30595; LF-111; 17β-Hydroxy-6β,7β:15β,16β-dimethylene-3-oxo-17α-pregn-4-ene-21-carboxylic acid, γ-lactone |
| AHFS/Drugs.com | Professional Drug Facts |
| License data |
|
| Routes of administration | By mouth[1] |
| Drug class | Progestogen; Progestin; Antimineralocorticoid; Steroidal antiandrogen |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 66–85%[1][4][5] |
| Protein binding | 95–97% (to albumin)[3][1][4] |
| Metabolism | Liver (mostly CYP450-independent (reduction, sulfation, and cleavage of lactone ring), some CYP3A4 contribution)[4][6][7][8] |
| Metabolites | • Drospirenone acid[3] • 4,5-Dihydrodrospirenone 3-sulfate[3] |
| Elimination half-life | 25–33 hours[3][4][1] |
| Excretion | Urine, feces[3] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.060.599 |
| Chemical and physical data | |
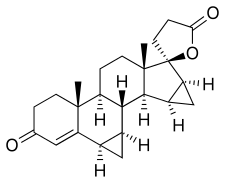
| Formula | C24H30O3 |
| Molar mass | 366.501 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Common side effects include acne, headache, breast tenderness, weight increase, and menstrual changes.[3] Rare side effects may include high potassium levels and blood clots (when taken as a combined oestrogen-progestogen pill), among others.[3][11] Drospirenone is a progestin, or a synthetic progestogen, and hence is an agonist of the progesterone receptor, the biological target of progestogens like progesterone.[1] It has additional antimineralocorticoid and antiandrogenic activity and no other important hormonal activity.[1] Because of its antimineralocorticoid activity and lack of undesirable off-target activity, drospirenone is said to more closely resemble bioidentical progesterone than other progestins.[12][13]
Drospirenone was patented in 1976 and introduced for medical use in 2000.[14][15] It is available widely throughout the world.[9] The medication is sometimes referred to as a "fourth-generation" progestin.[16][17] It is available as a generic medication.[18] In 2020, a formulation of drospirenone with ethinylestradiol was the 145th most commonly prescribed medication in the United States, with more than 4 million prescriptions.[19][20]