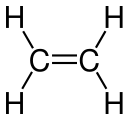
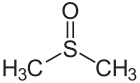
Double bond
Chemical bond involving four bonding electrons; has one sigma plus one pi bond / From Wikipedia, the free encyclopedia
In chemistry, a double bond is a covalent bond between two atoms involving four bonding electrons as opposed to two in a single bond. Double bonds occur most commonly between two carbon atoms, for example in alkenes. Many double bonds exist between two different elements: for example, in a carbonyl group between a carbon atom and an oxygen atom. Other common double bonds are found in azo compounds (N=N), imines (C=N), and sulfoxides (S=O). In a skeletal formula, a double bond is drawn as two parallel lines (=) between the two connected atoms; typographically, the equals sign is used for this.[1][2] Double bonds were introduced in chemical notation by Russian chemist Alexander Butlerov.[citation needed]
Double bonds involving carbon are stronger and shorter than single bonds. The bond order is two. Double bonds are also electron-rich, which makes them potentially more reactive in the presence of a strong electron acceptor (as in addition reactions of the halogens).
- Chemical compounds with double bonds
- Ethylene Carbon-carbon double bond
- Acetone Carbon-oxygen double bond
- Dimethyl sulfoxide Sulfur-oxygen double bond
- Diazene Nitrogen-nitrogen double bond