
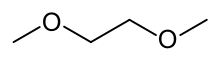
Dimethoxyethane
Chemical compound / From Wikipedia, the free encyclopedia
Not to be confused with dimethoxymethane or dimethyl ether.
Dimethoxyethane, also known as glyme, monoglyme, dimethyl glycol, ethylene glycol dimethyl ether, dimethyl cellosolve, and DME, is a colorless, aprotic, and liquid ether that is used as a solvent, especially in batteries.[2] Dimethoxyethane is miscible with water.
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
1,2-Dimethoxyethane[1] | |
| Other names
Ethane-1,2-diyl dimethyl ether[1] DME Glyme Ethylene glycol dimethyl ether Monoglyme Dimethyl glycol Dimethyl cellosolve | |
| Identifiers | |
| |
3D model (JSmol) |
|
| Abbreviations | DME |
| 1209237 | |
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.003.451 |
| EC Number |
|
| 1801 | |
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| C4H10O2 | |
| Molar mass | 90.122 g·mol−1 |
| Appearance | Colorless liquid |
| Density | 0.8683 g/cm3 |
| Melting point | −58 °C (−72 °F; 215 K) |
| Boiling point | 85 °C (185 °F; 358 K) |
| miscible | |
| Hazards | |
| GHS labelling: | |
   | |
| Danger | |
| H225, H332, H360FD | |
| P201, P202, P210, P233, P240, P241, P242, P243, P261, P271, P280, P281, P303+P361+P353, P304+P312, P304+P340, P308+P313, P312, P370+P378, P403+P235, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Flash point | −2 °C (28 °F; 271 K) |
| Related compounds | |
Related Ethers |
Dimethoxymethane |
Related compounds |
Ethylene glycol 1,4-Dioxane Diethylene glycol dimethyl ether |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
