
Beryllium oxide
Chemical compound / From Wikipedia, the free encyclopedia
Beryllium oxide (BeO), also known as beryllia, is an inorganic compound with the formula BeO. This colourless solid is a electrical insulator with a higher thermal conductivity than any other non-metal except diamond, and exceeds that of most metals.[12] As an amorphous solid, beryllium oxide is white. Its high melting point leads to its use as a refractory material.[13] It occurs in nature as the mineral bromellite. Historically and in materials science, beryllium oxide was called glucina or glucinium oxide, owing to its sweet taste.
Quick Facts Names, Identifiers ...
 | |
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Beryllium(II) monoxide | |
| Systematic IUPAC name
Oxoberyllium | |
| Other names
Beryllia, Thermalox, Bromellite, Thermalox 995.[1] | |
| Identifiers | |
3D model (JSmol) |
|
| 3902801 | |
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.013.758 |
| EC Number |
|
| MeSH | beryllium+oxide |
PubChem CID |
|
| RTECS number |
|
| UNII | |
| UN number | 1566 |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| BeO | |
| Molar mass | 25.011 g·mol−1 |
| Appearance | Colourless, vitreous crystals |
| Odor | Odourless |
| Density | 3.01 g/cm3[2] |
| Melting point | 2,578 °C (4,672 °F; 2,851 K)[2] |
| Band gap | 10.6 eV[3] |
| −11.9·10−6 cm3/mol[4] | |
| Thermal conductivity | 210 W/(m·K)[5] |
Refractive index (nD) |
n11.7184, n2=1.733[6][7] |
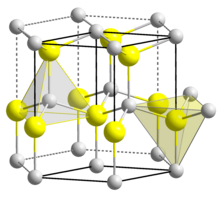
| Structure[8] | |
| Hexagonal, zincite | |
| P63mc | |
| C6v | |
a = 2.6979 Å, c = 4.3772 Å | |
Formula units (Z) |
2 |
| Linear | |
| Thermochemistry[9] | |
Heat capacity (C) |
25.6 J/(K·mol) |
Std molar entropy (S⦵298) |
13.77±0.04 J/(K·mol) |
Std enthalpy of formation (ΔfH⦵298) |
−609.4±2.5 kJ/mol |
Gibbs free energy (ΔfG⦵) |
−580.1 kJ/mol |
Enthalpy of fusion (ΔfH⦵fus) |
86 kJ/mol |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Very toxic, Group 1B carcinogen |
| GHS labelling: | |
   | |
| Danger | |
| H301, H315, H317, H319, H330, H335, H350, H372 | |
| P201, P260, P280, P284, P301+P310, P305+P351+P338 | |
| NFPA 704 (fire diamond) | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
15 mg/kg (mouse, oral)[10] |
| NIOSH (US health exposure limits): | |
PEL (Permissible) |
TWA 0.002 mg/m3 C 0.005 mg/m3 (30 minutes), with a maximum peak of 0.025 mg/m3 (as Be)[11] |
REL (Recommended) |
Ca C 0.0005 mg/m3 (as Be)[11] |
IDLH (Immediate danger) |
Ca [4 mg/m3 (as Be)][11] |
| Related compounds | |
Other anions |
Beryllium telluride |
Other cations |
|
| Supplementary data page | |
| Beryllium oxide (data page) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close
