
Aqua regia
Mixture of nitric acid and hydrochloric acid in a 1:3 molar ratio / From Wikipedia, the free encyclopedia
"🜇" redirects here. For the numismatic abbreviation, see List of numismatic abbreviations.
For the song by Sleep Token, see Take Me Back to Eden.
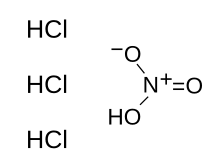
Aqua regia (/ˈreɪɡiə, ˈriːdʒiə/; from Latin, "regal water" or "royal water") is a mixture of nitric acid and hydrochloric acid, optimally in a molar ratio of 1:3.[lower-alpha 2] Aqua regia is a fuming liquid. Freshly prepared aqua regia is colorless, but it turns yellow, orange or red within seconds from the formation of nitrosyl chloride and nitrogen dioxide. It was so named by alchemists because it can dissolve noble metals like gold and platinum, though not all metals.
Quick Facts Names, Identifiers ...
 | |
| Names | |
|---|---|
| IUPAC name
Nitric acid trihydrochloride | |
Other names
| |
| Identifiers | |
3D model (JSmol) |
|
PubChem CID |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| Properties | |
| HNO3 + 3 HCl | |
| Appearance | Fuming liquid. Freshly prepared aqua regia is colorless, but it turns yellow, orange or red within seconds. |
| Density | 1.01–1.21 g/cm3 |
| Melting point | −42 °C (−44 °F; 231 K) |
| Boiling point | 108 °C (226 °F; 381 K) |
| Miscible | |
| Vapor pressure | 21 mbar |
| Hazards | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close


