
Anode
Electrode through which conventional current flows into a polarized electrical device / From Wikipedia, the free encyclopedia
An anode is an electrode of a polarized electrical device through which conventional current enters the device. This contrasts with a cathode, an electrode of the device through which conventional current leaves the device. A common mnemonic is ACID, for "anode current into device".[1] The direction of conventional current (the flow of positive charges) in a circuit is opposite to the direction of electron flow, so (negatively charged) electrons flow from the anode of a galvanic cell, into an outside or external circuit connected to the cell. For example, the end of a household battery marked with a "+" is the cathode (while discharging).
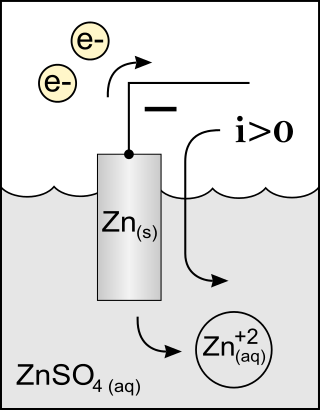
In both a galvanic cell and an electrolytic cell, the anode is the electrode at which the oxidation reaction occurs. In a galvanic cell the anode is the wire or plate having excess negative charge as a result of the oxidation reaction. In an electrolytic cell, the anode is the wire or plate upon which excess positive charge is imposed.[2] As a result of this, anions will tend to move towards the anode where they will undergo oxidation.
Historically, the anode of a galvanic cell was also known as the zincode because it was usually composed of zinc.[3][4]: pg. 209, 214