
Aminopenicillin
Group of antibiotics / From Wikipedia, the free encyclopedia
The aminopenicillins are a group of antibiotics in the penicillin family that are structural analogs of ampicillin (which is the 2-amino derivative of benzylpenicillin, hence the name).[1] Like other penicillins and beta-lactam antibiotics, they contain a beta-lactam ring that is crucial to its antibacterial activity.[citation needed]
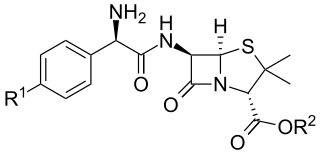

Aminopenicillins feature a positively charged amino group that enhances their uptake through bacterial porin channels. This does not, however, prevent resistance conferred by bacterial beta-lactamases.[2] Members of this family include ampicillin, amoxicillin and bacampicillin.[3]