
Tin selenide
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Tin selenide?
Summarize this article for a 10 year old
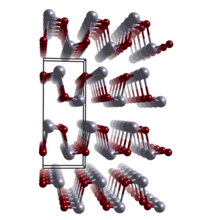
Tin selenide, also known as stannous selenide, is an inorganic compound with the formula SnSe. Tin(II) selenide is a typical layered metal chalcogenide[4] as it includes a group 16 anion (Se2−) and an electropositive element (Sn2+), and is arranged in a layered structure. Tin(II) selenide is a narrow band-gap (IV-VI) semiconductor structurally analogous to black phosphorus. It has received considerable interest for applications including low-cost photovoltaics, and memory-switching devices.
 | |
| Names | |
|---|---|
| Other names
Tin(II) selenide | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.013.871 |
| EC Number |
|
PubChem CID |
|
| UNII | |
| |
| |
| Properties | |
| SnSe | |
| Molar mass | 197.67 g/mol |
| Appearance | steel gray odorless powder |
| Density | 5.75 g/cm3[1] |
| Melting point | 861 °C (1,582 °F; 1,134 K) |
| negligible | |
| Band gap | 0.9 eV (indirect), 1.3 eV (direct)[2] |
| Structure | |
| Orthorhombic, oP8[2] | |
| Pnma, No. 62[2] | |
| Thermochemistry | |
Std enthalpy of formation (ΔfH⦵298) |
-88.7 kJ/mol |
| Hazards | |
| GHS labelling: | |
   | |
| Danger | |
| H301, H331, H373, H410 | |
| P260, P261, P264, P270, P271, P273, P301+P310, P304+P340, P311, P314, P321, P330, P391, P403+P233, P405, P501 | |
| NFPA 704 (fire diamond) | |
| Safety data sheet (SDS) | https://www.ltschem.com/msds/SnSe.pdf |
| Related compounds | |
Other anions |
Tin(II) oxide Tin(II) sulfide Tin telluride |
Other cations |
Carbon monoselenide Silicon monoselenide Germanium selenide Lead selenide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Because of its low thermal conductivity as well as reasonable electrical conductivity, tin selenide is one of the most efficient thermoelectric materials.[5][6]
