
Silylgermane
Chemical compound / From Wikipedia, the free encyclopedia
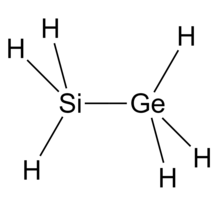
Silylgermane is an inorganic compound with the chemical formula H3Si−GeH3. It is a colorless gas with an unpleasant odor. It is unstable in air. It is very flammable, very toxic and corrosive. It reacts with alkali liberating hydrogen.[2][5][4][3]
Quick Facts Names, Identifiers ...
 | |
| Names | |
|---|---|
| IUPAC name
Silylgermane | |
| Other names | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| EC Number |
|
PubChem CID |
|
| |
| |
| Properties | |
| SiH3GeH3 | |
| Molar mass | 106.763 g·mol−1 |
| Appearance | Colorless gas |
| Odor | Unpleasant, irritating[3] |
| Melting point | −119.7 °C (−183.5 °F; 153.5 K)[4] |
| Boiling point | 7.0 °C (44.6 °F; 280.1 K)[4] |
| Structure | |
| Ethane-like | |
| Hazards[2] | |
| Occupational safety and health (OHS/OSH): | |
Main hazards |
Extremely flammable, toxic and corrosive, may cause severe and permanent eye damage, fatal if inhaled |
Inhalation hazards |
Fatal |
Eye hazards |
Permanent eye damage |
Skin hazards |
Corrosive injuries |
| GHS labelling: | |
    | |
| Danger | |
| H220, H280, H314, H330 | |
| P210, P222, P230, P260, P264, P271, P280, P284, P301+P330+P331, P302, P304+P340, P305, P316, P320, P321, P338, P361, P363, P377, P381, P403, P403+P233, P405, P410+P403, P501 | |
| Related compounds | |
Related compounds |
|
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Close