
Octreotide
Octapeptide that mimics natural somatostatin pharmacologically / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Octreotide?
Summarize this article for a 10 year old
Octreotide, sold under the brand name Sandostatin among others, is an octapeptide that mimics natural somatostatin pharmacologically, though it is a more potent inhibitor of growth hormone, glucagon, and insulin than the natural hormone. It was first synthesized in 1979 by the chemist Wilfried Bauer, and binds predominantly to the somatostatin receptors SSTR2 and SSTR5.[4]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Sandostatin, Bynfezia Pen, Mycapssa, others |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a693049 |
| License data |
|
| Pregnancy category |
|
| Routes of administration | Subcutaneous, intramuscular, intravenous, by mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 60% (IM), 100% (SC) |
| Protein binding | 40–65% |
| Metabolism | Liver |
| Elimination half-life | 1.7–1.9 hours |
| Excretion | Urine (32%) |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID |
|
| IUPHAR/BPS | |
| DrugBank |
|
| ChemSpider | |
| UNII |
|
| KEGG | |
| ChEBI | |
| ChEMBL |
|
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
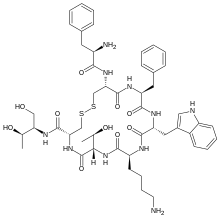
| Formula | C49H66N10O10S2 |
| Molar mass | 1019.25 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
It was approved for use in the United States in 1988.[2][1] Octreotide (Mycapssa) was approved for medical use in the European Union in 2022.[3] As of June 2020[update], octreotide (Mycapssa) is the first and only oral somatostatin analog (SSA) approved by the FDA.[5] It is on the World Health Organization's List of Essential Medicines.[6]