
Live attenuated influenza vaccine
Nasal influenza vaccine / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Live attenuated influenza vaccine?
Summarize this article for a 10 year old
Live attenuated influenza vaccine (LAIV) is a type of influenza vaccine in the form of a nasal spray that is recommended for the prevention of influenza. It was developed by the Syrian-American epidemiologist Hunein Maassab.[5]
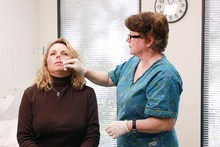 Nurse administering the FluMist product | |
| Vaccine description | |
|---|---|
| Target | Influenza |
| Vaccine type | Attenuated |
| Clinical data | |
| Trade names | Flumist Quadrivalent, Fluenz Tetra |
| AHFS/Drugs.com | Monograph |
| License data | |
| Routes of administration | Intranasal |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| CAS Number | |
| ChemSpider |
|
| KEGG | |
| | |
It is an attenuated live vaccine, unlike other influenza vaccines, which are inactivated vaccines. Live attenuated influenza vaccine is administered intranasally,[6] while inactivated vaccines are administered by intramuscular injection. Live attenuated influenza vaccine is sold under the brand name FluMist Quadrivalent in the United States and the brand name Fluenz Tetra in the European Union.[7][4] FluMist was first introduced in 2003 by MedImmune.[8][9][10]