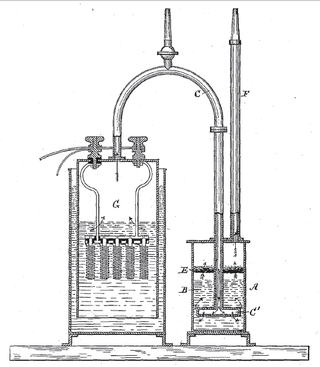
Electrolytic cell
Cell that uses electrical energy to drive a non-spontaneous redox reaction / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Electrolytic cell?
Summarize this article for a 10 year old
An electrolytic cell is an electrochemical cell that utilizes an external source of electrical energy to force a chemical reaction that would otherwise not occur.[1]: 64, 89 [2]: GL7 The external energy source is a voltage applied between the cell's two electrodes; an anode (positively charged electrode) and a cathode (negatively charged electrode), which are immersed in an electrolyte solution.[1]: 89 [3][page needed] This is in contrast to a galvanic cell, which itself is a source of electrical energy and the foundation of a battery.[1]: 64 The net reaction taking place in a galvanic cell is a spontaneous reaction, i.e., the Gibbs free energy remains -ve, while the net reaction taking place in an electrolytic cell is the reverse of this spontaneous reaction, i.e., the Gibbs free energy is +ve.[3][page needed]
This article possibly contains original research. (November 2017) |
This article needs additional citations for verification. (November 2023) |