
Chlortetracycline
Chemical compound / From Wikipedia, the free encyclopedia
Dear Wikiwand AI, let's keep it short by simply answering these key questions:
Can you list the top facts and stats about Chlortetracycline?
Summarize this article for a 10 year old
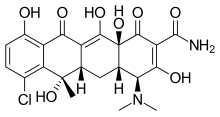
Chlortetracycline (trade name Aureomycin, Lederle Laboratories) is a tetracycline antibiotic, the first tetracycline to be identified. It was discovered in 1945 at Lederle Laboratories under the supervision of scientist Yellapragada Subbarow, Benjamin Minge Duggar. They were helped by Louis T. Wright,[2] a surgeon who conducted this medications first human experiments. Duggar identified the antibiotic as the product of an actinomycete he cultured from a soil sample collected from Sanborn Field at the University of Missouri.[3] The organism was named Streptomyces aureofaciens and the isolated drug, Aureomycin, because of their golden color.[1]
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Aureomycin |
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| Routes of administration | By mouth, IV, topical |
| ATC code | |
| Pharmacokinetic data | |
| Bioavailability | 30% |
| Protein binding | 50 to 55% |
| Metabolism | Gastrointestinal tract, hepatic (75%) |
| Metabolites | Isochlortetracycline |
| Elimination half-life | 5.6 to 9 hours |
| Excretion | 60% renal and >10% biliary |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| DrugBank |
|
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEMBL | |
| E number | E702 (antibiotics) |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.000.310 |
| Chemical and physical data | |
| Formula | C22H23ClN2O8 |
| Molar mass | 478.88 g·mol−1 |
| 3D model (JSmol) | |
| Specific rotation | [α]D25−275°·cm3·dm−1·g−1 (methane) |
| Melting point | 168 to 169 °C (334 to 336 °F) |
| Solubility in water | 0.5–0.6 mg/mL (20 °C) |
| |
| |
| | |
It is on the World Health Organization's List of Essential Medicines.[4]