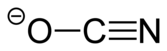氰酸銀是一種化合物,氰酸的銀鹽。它可以通過氰酸鉀或尿素與硝酸銀的反應來製備。[2]
| 氰酸銀 | |||
|---|---|---|---|
| |||
| 系統名 Silver(I) cyanate | |||
| 識別 | |||
| CAS號 | 3315-16-0 | ||
| PubChem | 76827 | ||
| ChemSpider | 69282 | ||
| SMILES |
| ||
| InChI |
| ||
| InChIKey | DWBPIWPCOSHWCK-REWHXWOFAQ | ||
| 性質 | |||
| 化學式 | AgOCN | ||
| 摩爾質量 | 149.885 g/mol g·mol⁻¹ | ||
| 危險性[1] | |||
| 歐盟危險性符號 | |||
| 警示術語 | R:R20/21/22 | ||
| 安全術語 | S:S24/25 | ||
| GHS危險性符號 | |||
| H-術語 | H302, H312, H332 | ||
| P-術語 | P280 | ||
| 若非註明,所有數據均出自標準狀態(25 ℃,100 kPa)下。 | |||
氰酸銀是一種米黃色到灰色的粉末。結晶為單斜,空間群P21/m,其中a = 547.3 pm,b = 637.2 pm,c = 341.6 pm,β = 91°。[3]
參見
參考
Wikiwand in your browser!
Seamless Wikipedia browsing. On steroids.
Every time you click a link to Wikipedia, Wiktionary or Wikiquote in your browser's search results, it will show the modern Wikiwand interface.
Wikiwand extension is a five stars, simple, with minimum permission required to keep your browsing private, safe and transparent.